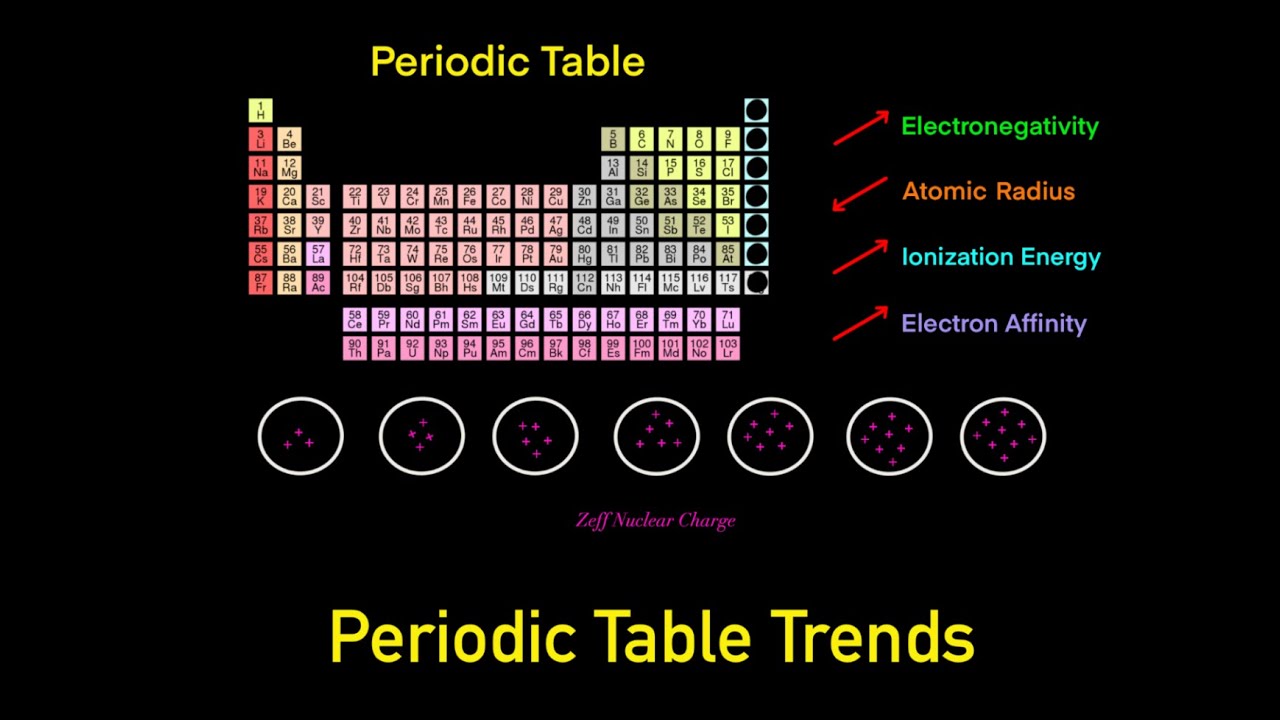
Periodic Table Trends Trick (Electronegativity, Atomic Radius, Ionization Energy, Electron Affinity) - YouTube

Ionization Energy: Periodic Table Trends | What is Ionization Energy? - Video & Lesson Transcript | Study.com
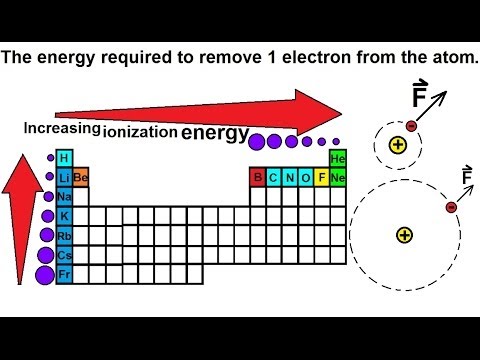
Chemistry - Periodic Variations (13 of 23) Atomic Radius: Ionization Energy and the Period Table - YouTube
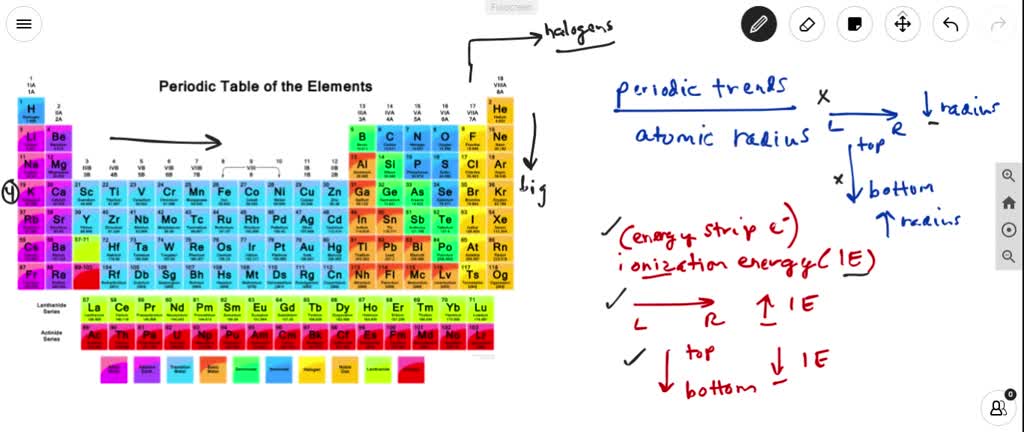
SOLVED:(a) What is the trend in first ionization energies as one proceeds down the group 17 elements? Explain how this trend relates to the variation in atomic radii. (b) What is the

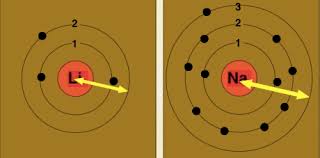
inorganic chemistry - Why does the ionization enthalpy of elements across a period not follow a regular pattern while the atomic size always decreases? - Chemistry Stack Exchange










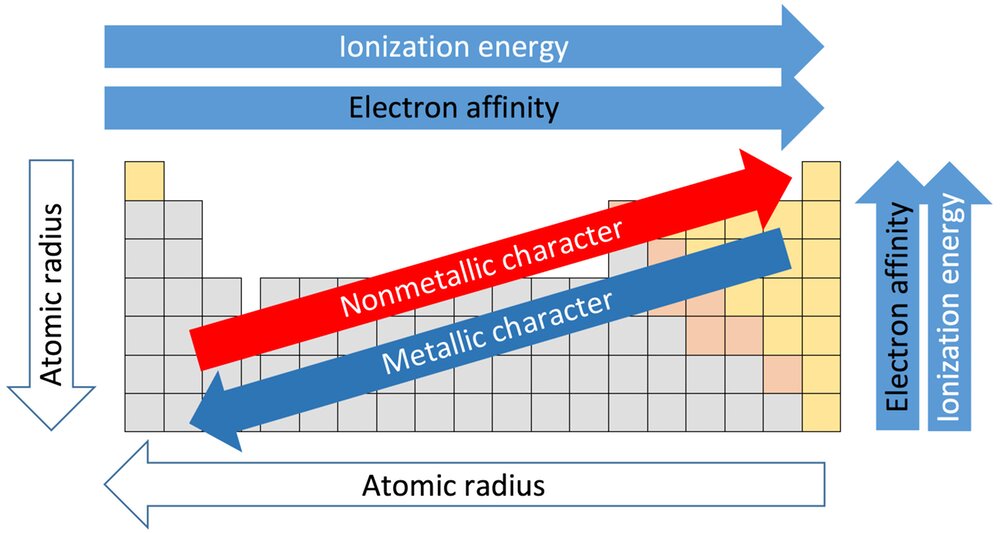
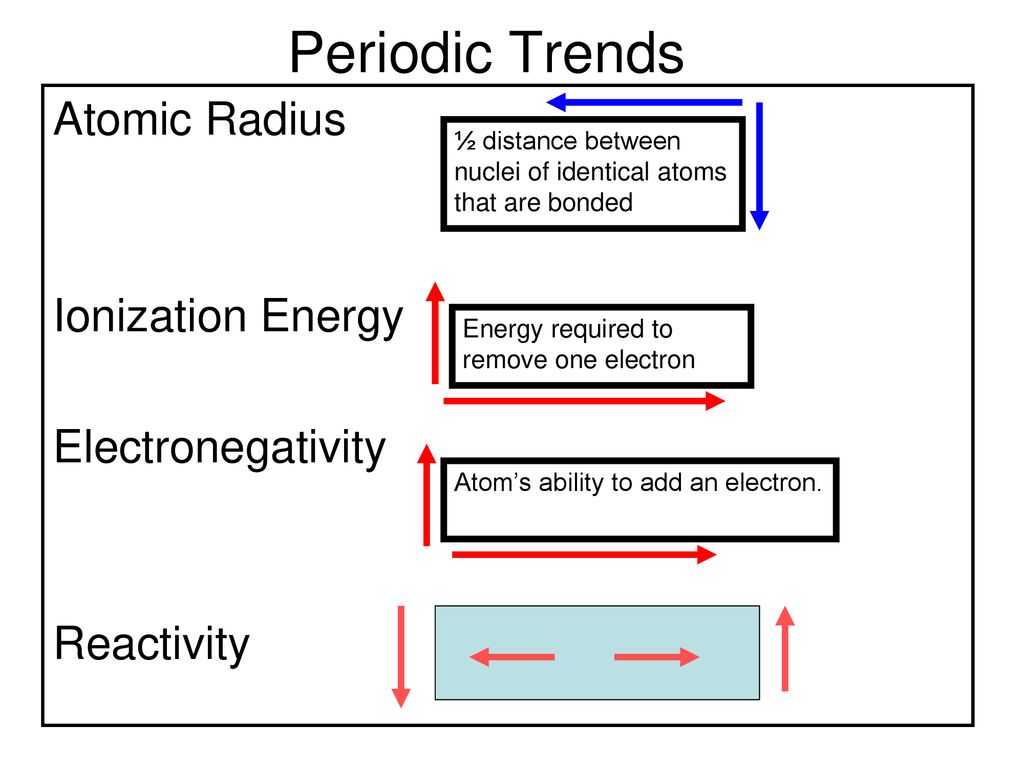



:max_bytes(150000):strip_icc()/chart-of-periodic-table-trends-608792-v1-6ee35b80170349e8ab67865a2fdfaceb.png)
