
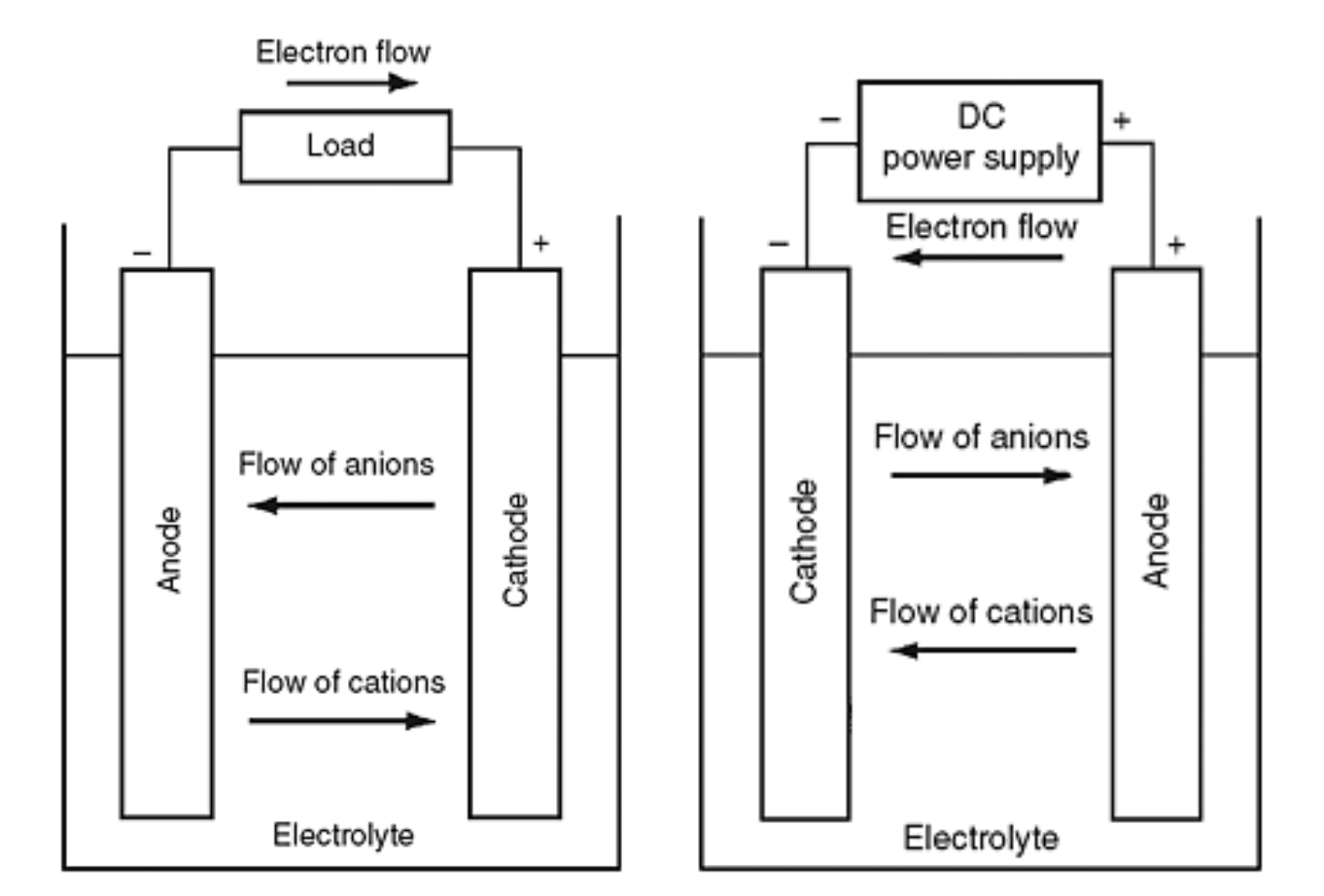
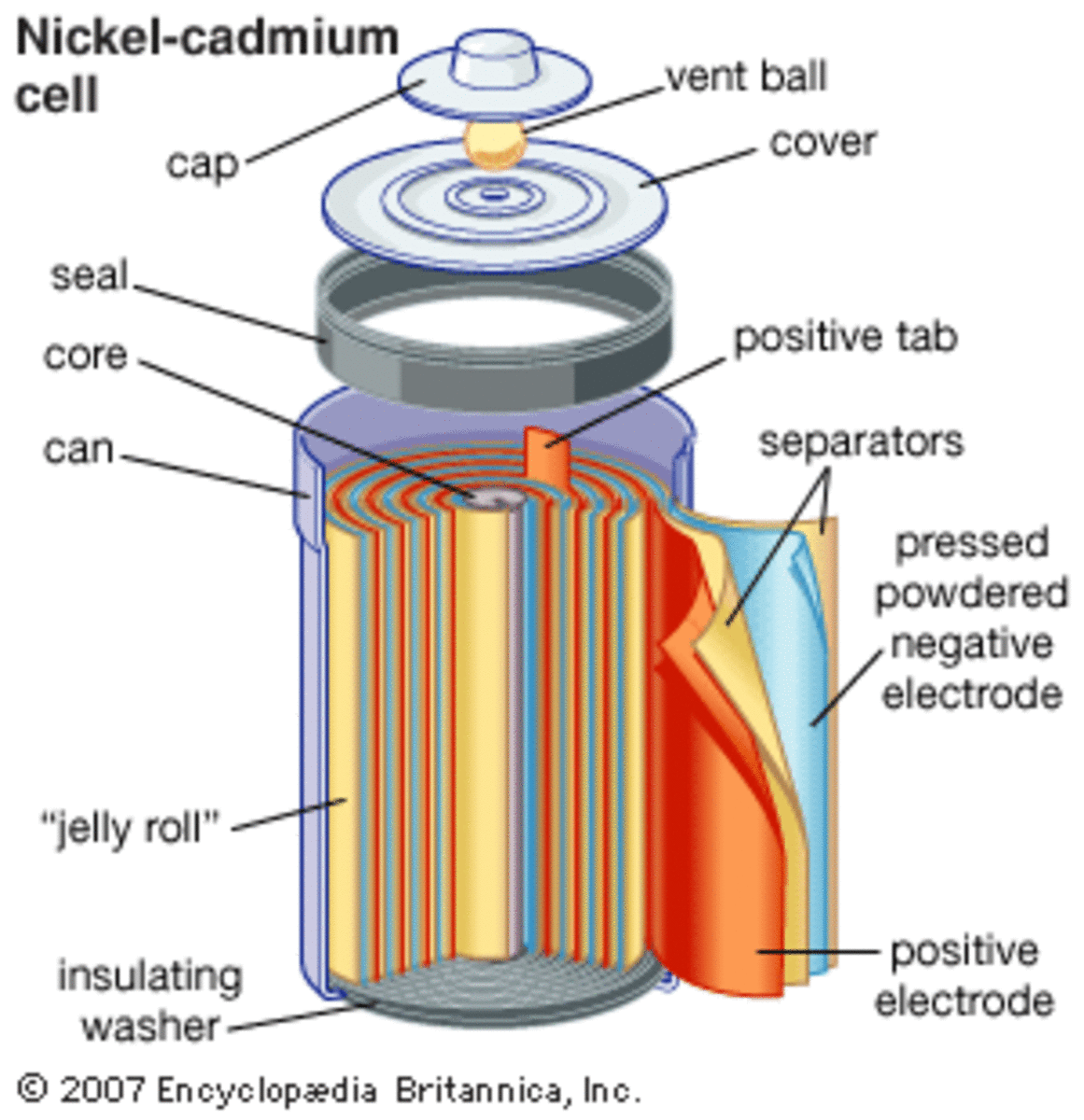
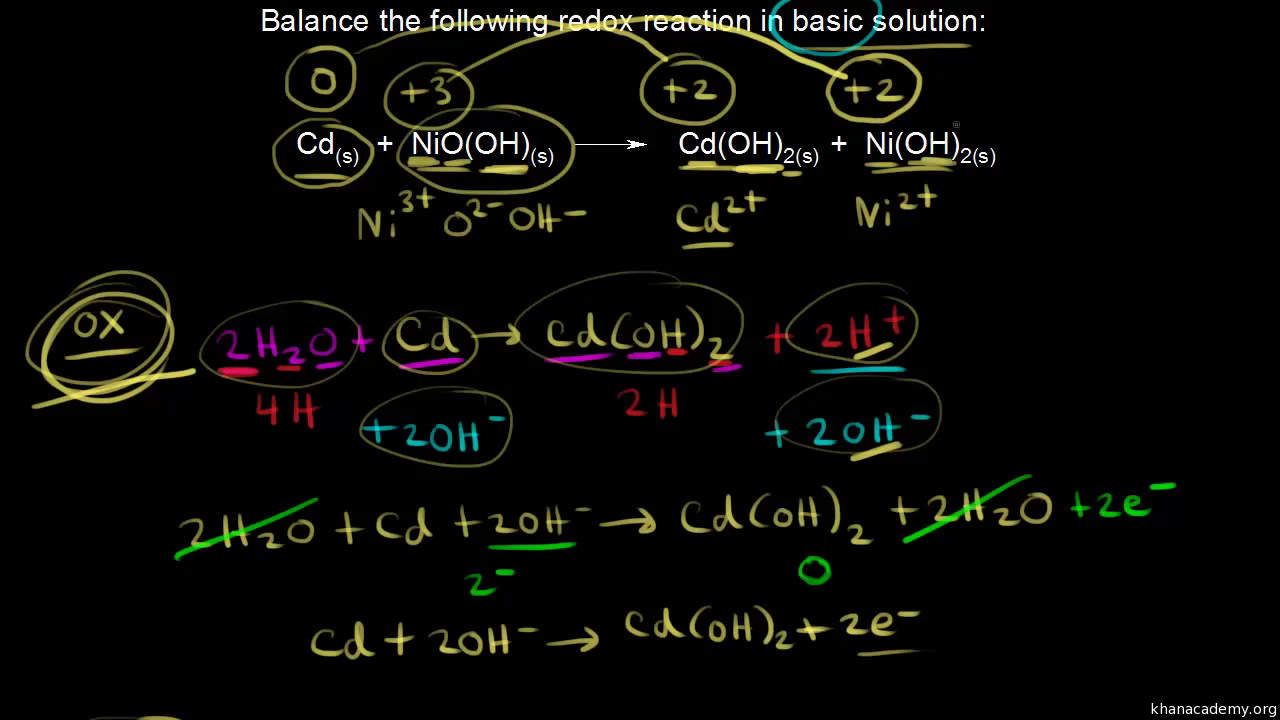
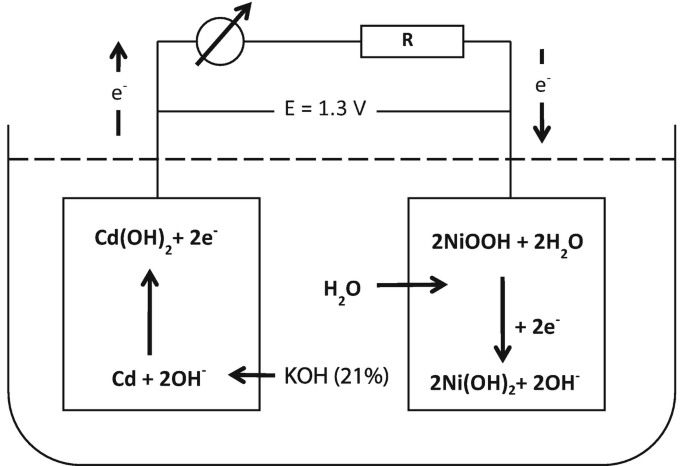
SOLVED: Nickel-Cadmium rechargeable batteries employ the following half- reactions Given the standard reduction potentials: Cd(OH)2(s) + 20 Cd(s) 2 OH-(aq) E" = -0.76 V NiO(OH)(s) Hzo( + € Ni(OH)2(s) OH-(aq) E' = +0.49

SOLVED: 2 Nickel - cadmium battery The reversible reactions involved in the chargeldischarge of a nickel-cadmium cell are: Cadmium electrode: Cd(OH)z + 2 e Cd + 2 OH AredG" =+156.3 kJlmol Nickel

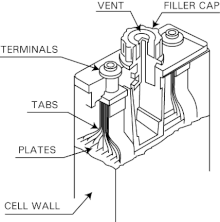





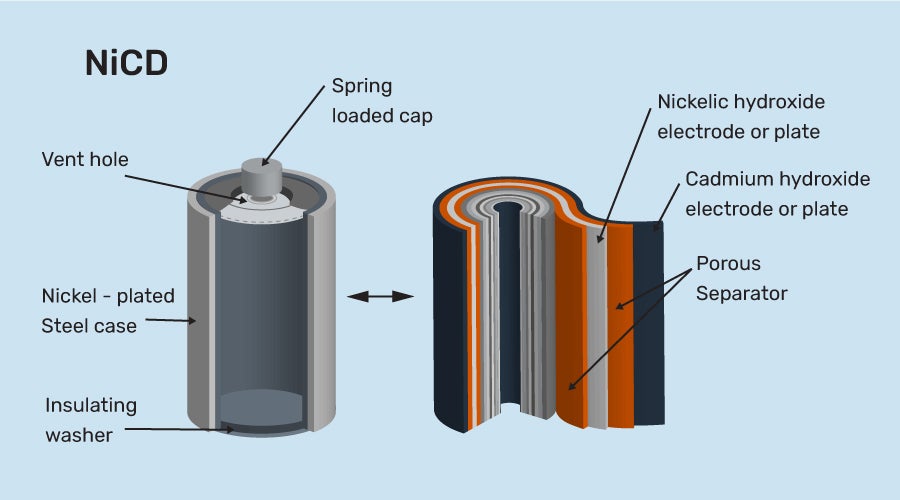
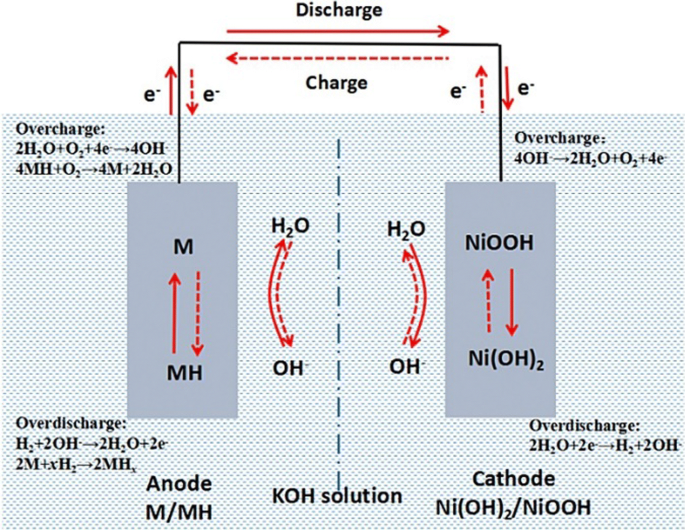




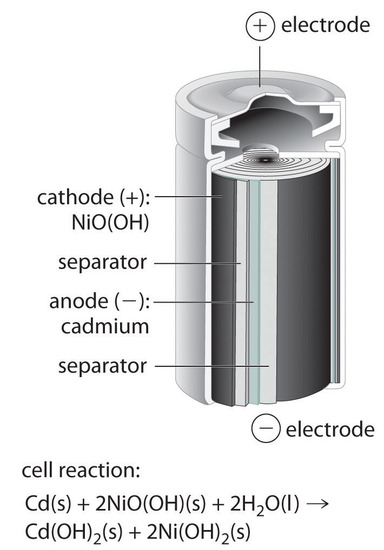

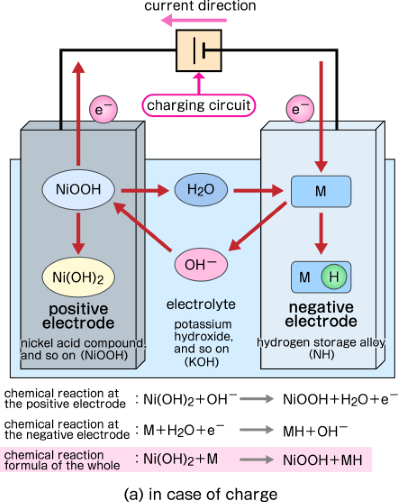
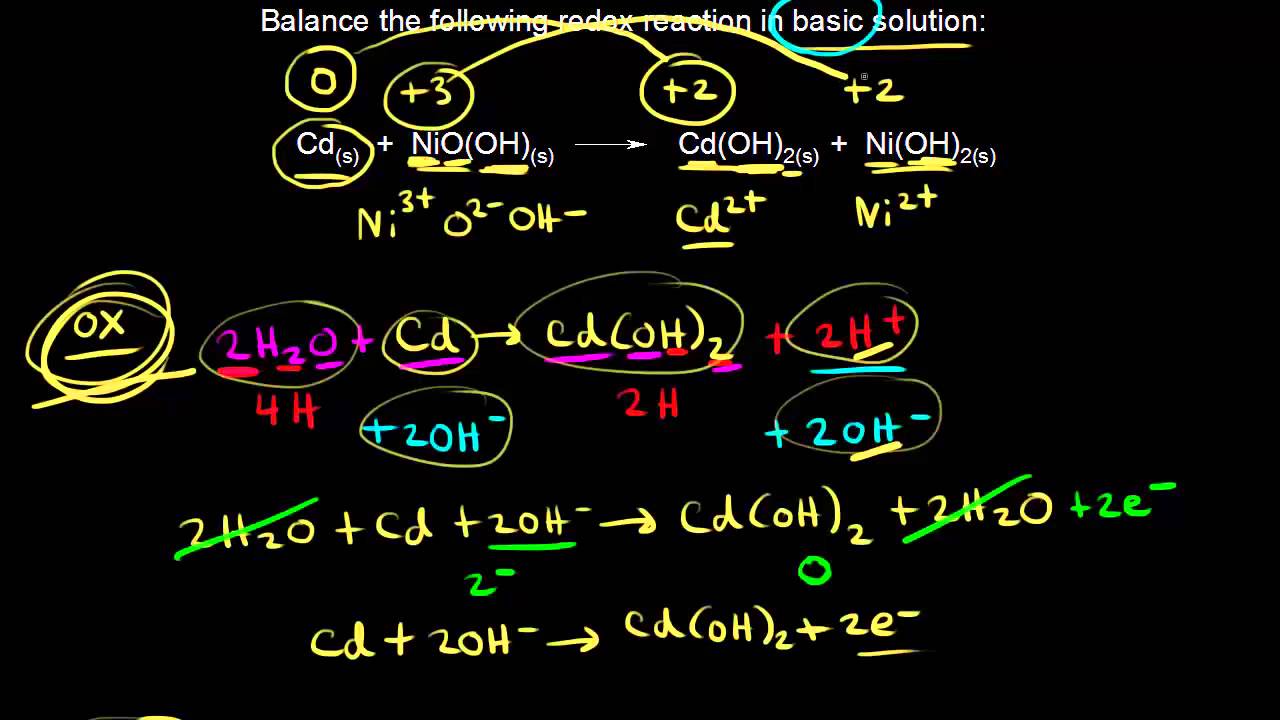
![C6 NiCd Rechargeable Battery [HL IB Chemistry] - YouTube C6 NiCd Rechargeable Battery [HL IB Chemistry] - YouTube](https://i.ytimg.com/vi/PINXU050LUM/maxresdefault.jpg)