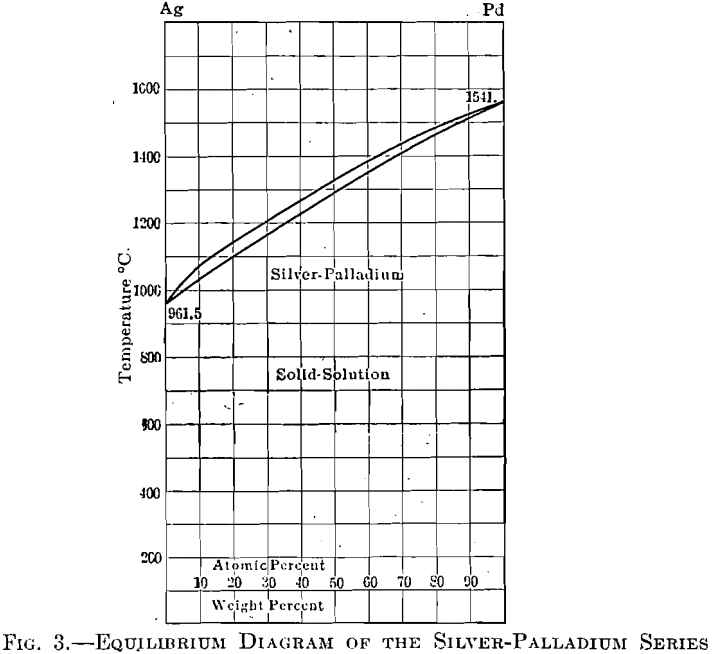
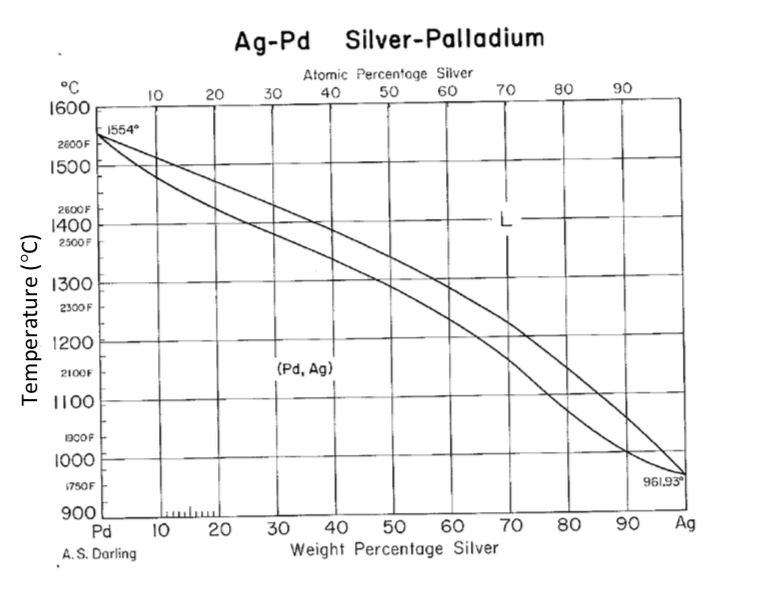
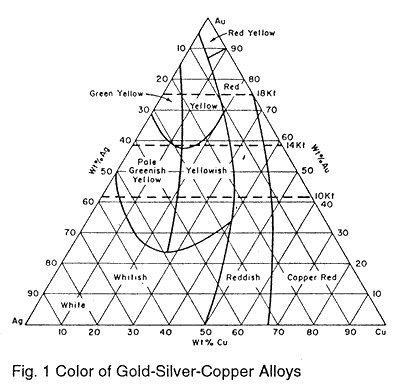
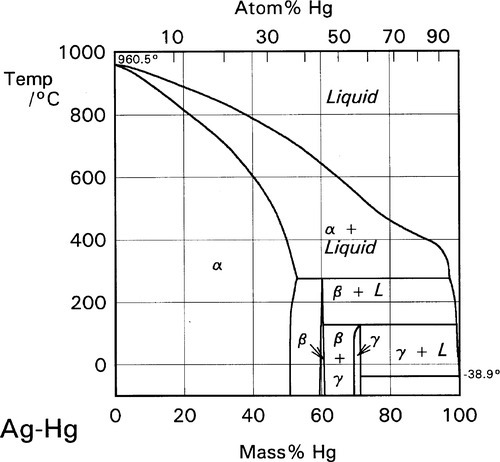
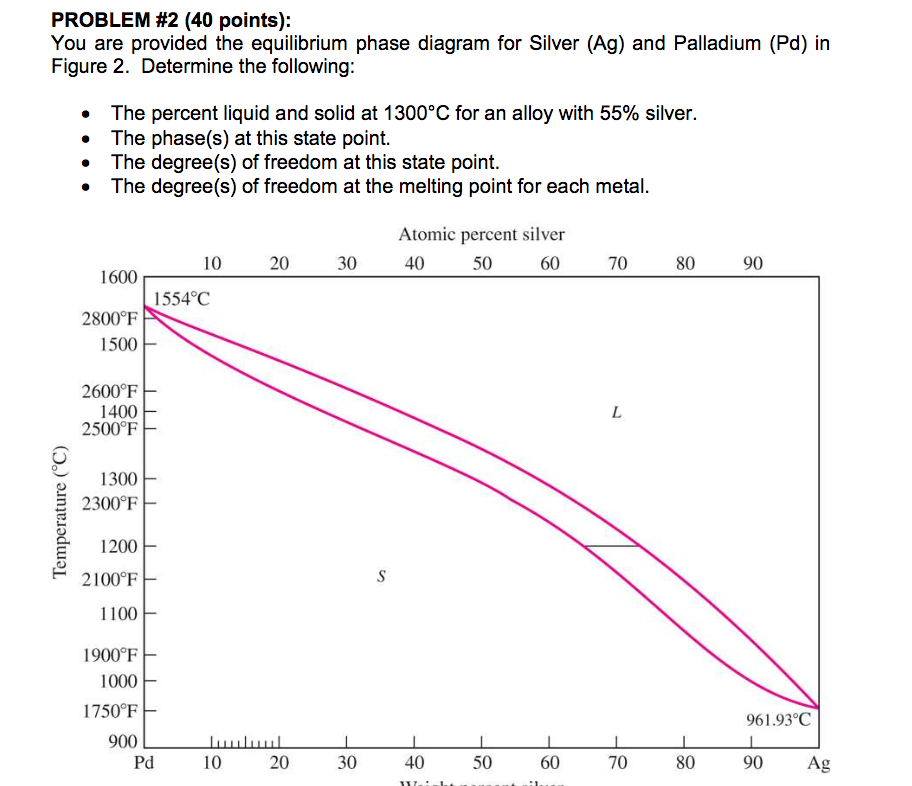
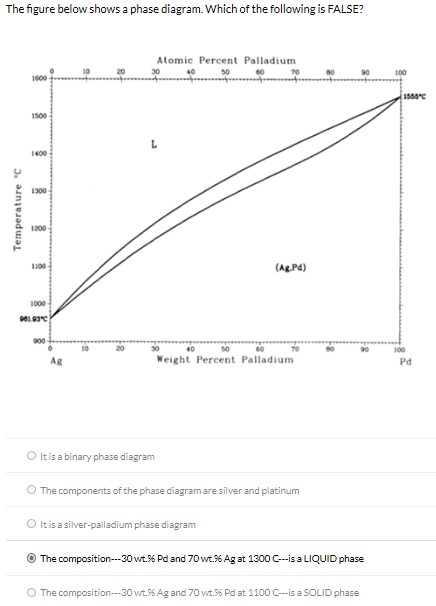
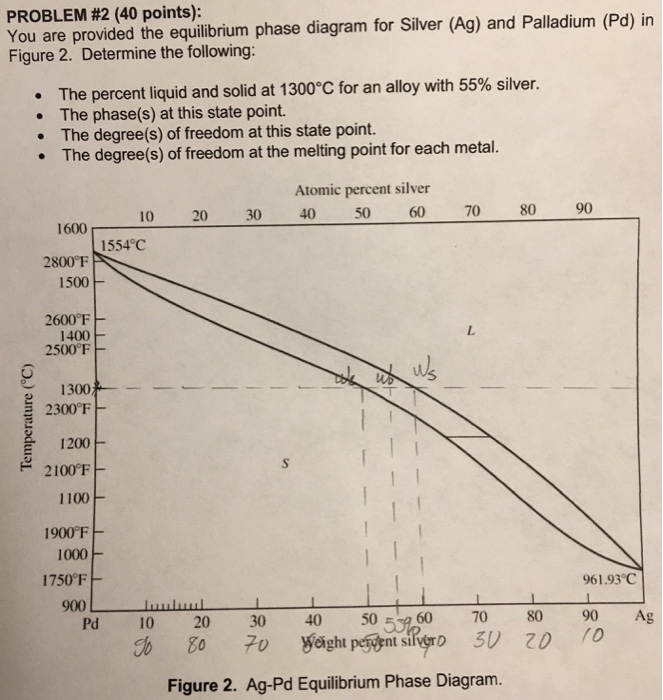
Tensile Properties of a Series of Palladium-Silver Alloys Exposed to Hydrogen | Journal of Energy Engineering | Vol 141, No 3
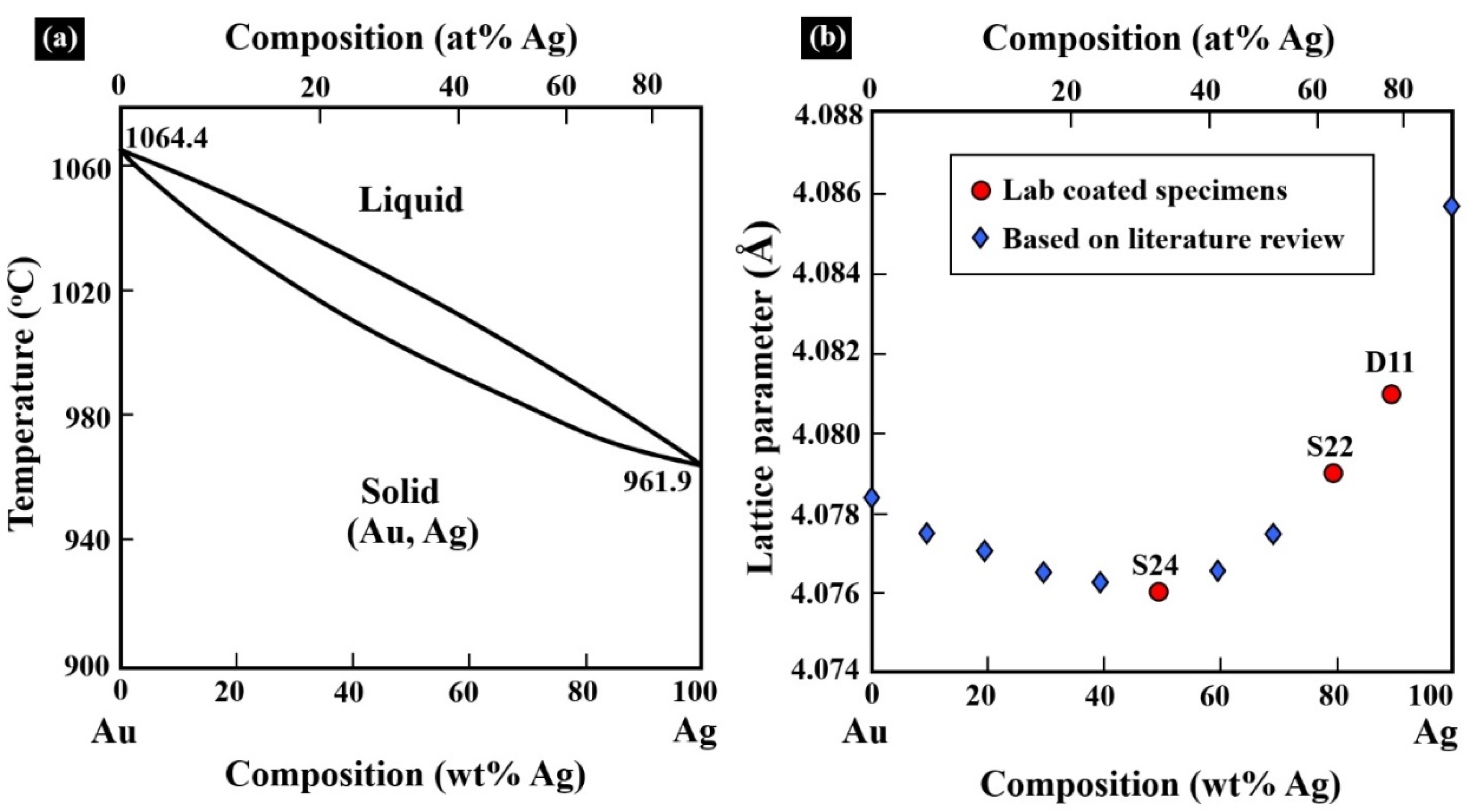
Metals | Free Full-Text | Gold–Silver Electroless Plating on Laser Powder-Bed Fusion Additively Printed AlSi10Mg Parts
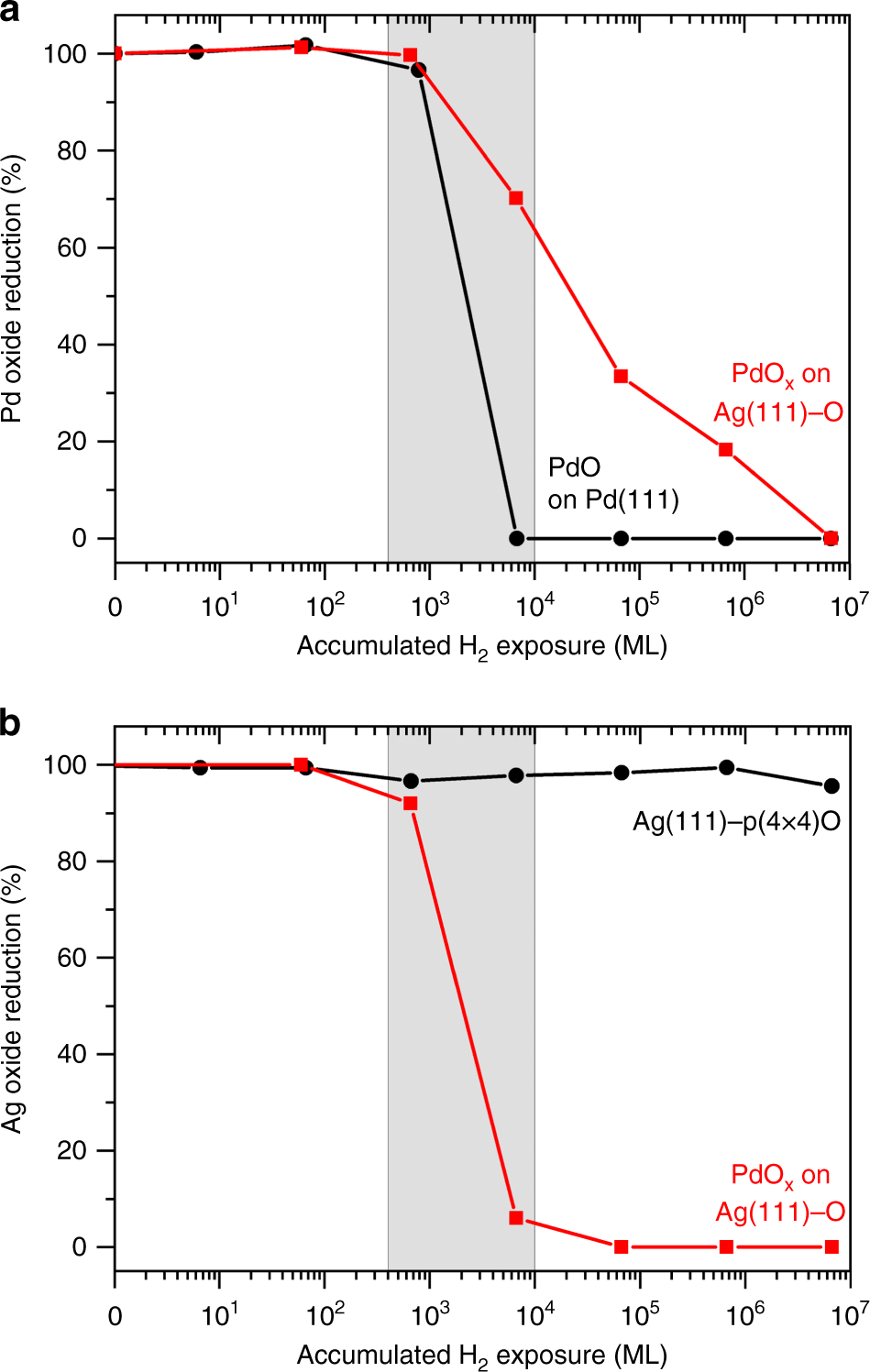
Hydrogen migration at restructuring palladium–silver oxide boundaries dramatically enhances reduction rate of silver oxide | Nature Communications

Tensile Properties of a Series of Palladium-Silver Alloys Exposed to Hydrogen | Journal of Energy Engineering | Vol 141, No 3
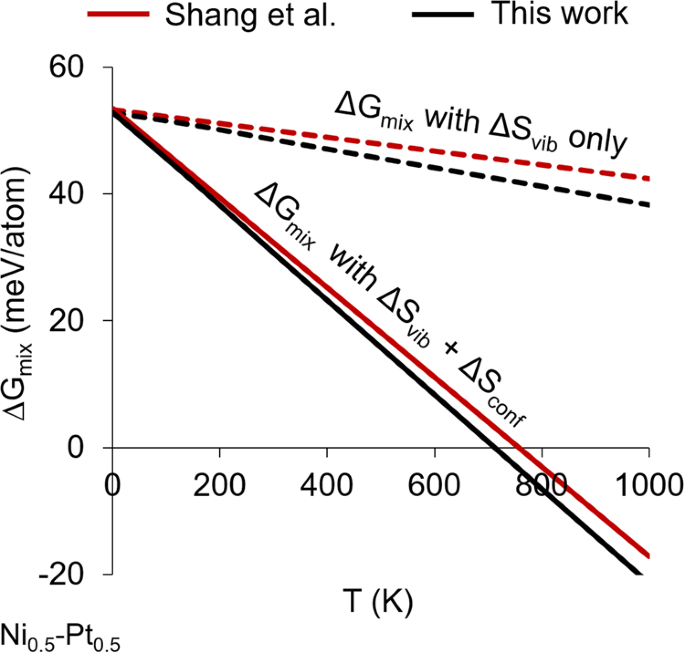
Entropy contributions to phase stability in binary random solid solutions | npj Computational Materials

The calculated Ag-Pd phase diagram with experimental points (Ref 4, 5) | Download Scientific Diagram
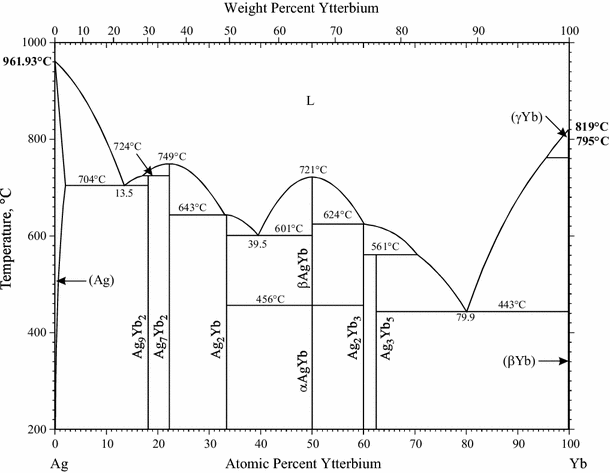
Supplemental Literature Review of Binary Phase Diagrams: Ag-Yb, Al-Co, Al-I, Co-Cr, Cs-Te, In-Sr, Mg-Tl, Mn-Pd, Mo-O, Mo-Re, Ni-Os, and V-Zr | SpringerLink

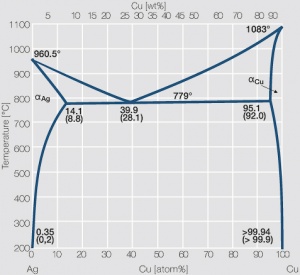
At 500 o C, what is the maximum solubility of (a) Cu dissolved in a matrix of Ag? (b) Ag dissolved in a matrix of Cu? For both, use units of wt%
What metal is easiest to alloy with Palladium and can it form a eutectic lowering the alloyed melting point? - Quora
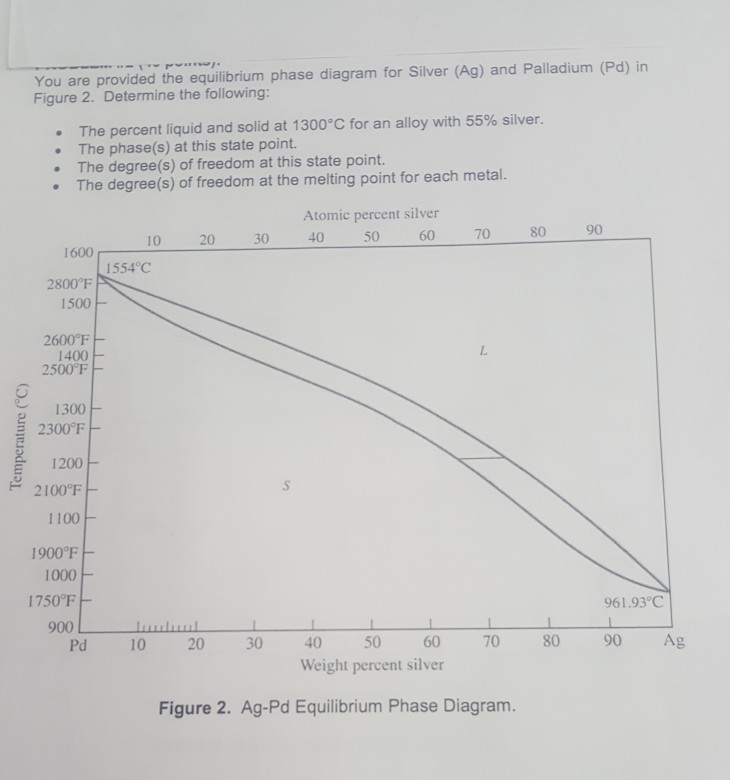
4: The phase diagram for Ag and Pd. The "'L"' signifies the aqueous phase. | Download Scientific Diagram