
SpringWorks on X: "We are happy to have initiated the rolling submission of our New Drug Application with the FDA for our investigational MEK inhibitor in pediatric and adult patients with neurofibromatosis

Pfizer Inc. on X: "BREAKING: Today, with @BioNTech_Group, we initiated the submission of a BLA for @FDA approval of the Pfizer-BioNTech #COVID19 vaccine to prevent COVID-19 in individuals 16 years and older.
Legend Biotech, J&J finish off rolling submission for CAR-T hopeful, approach finish line | Fierce Biotech

Santhera and ReveraGen Complete NDA Submission to FDA for Vamorolone in Duchenne Muscular Dystrophy - CureDuchenne

FA Research Alliance on X: "Reata Pharmaceuticals Completes Rolling Submission of New Drug Application for Omaveloxolone for the Treatment of Patients with Friedreich's Ataxia Click here to read the full press release:
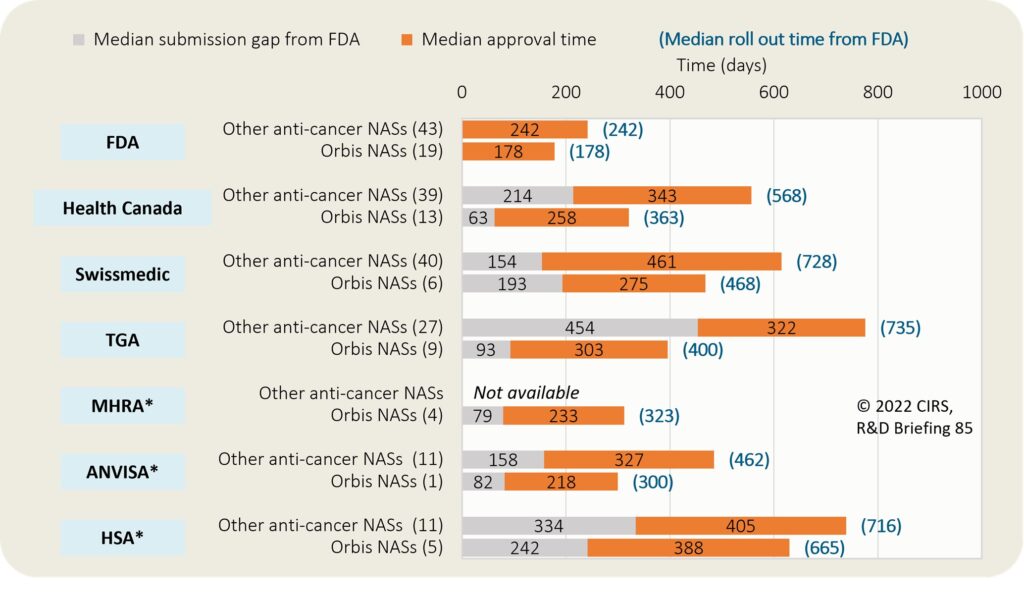
Shorter timelines, evolving strategies: Four key trends in regulatory approvals of new medicines - Clarivate


















