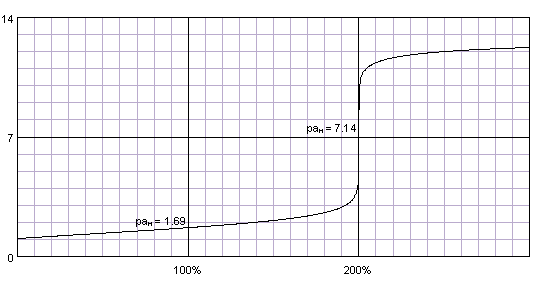
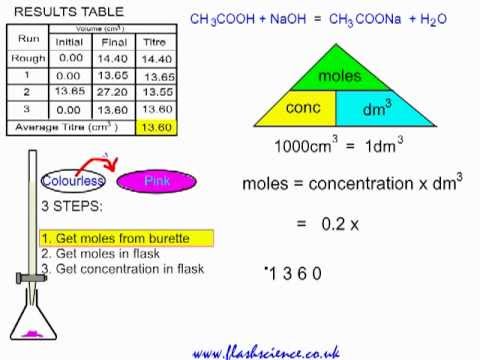
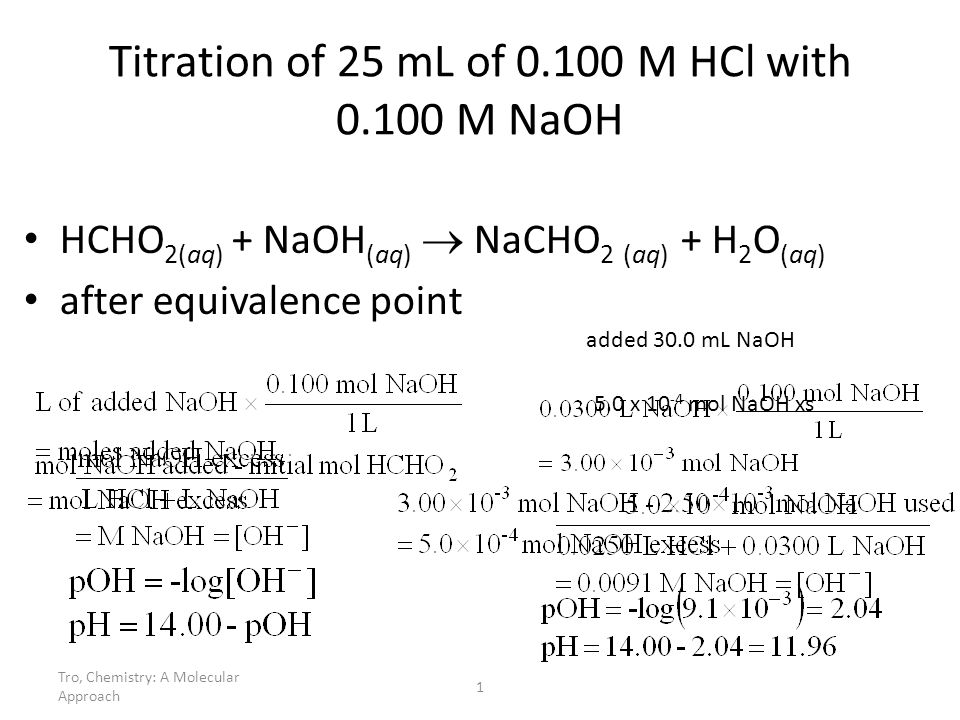
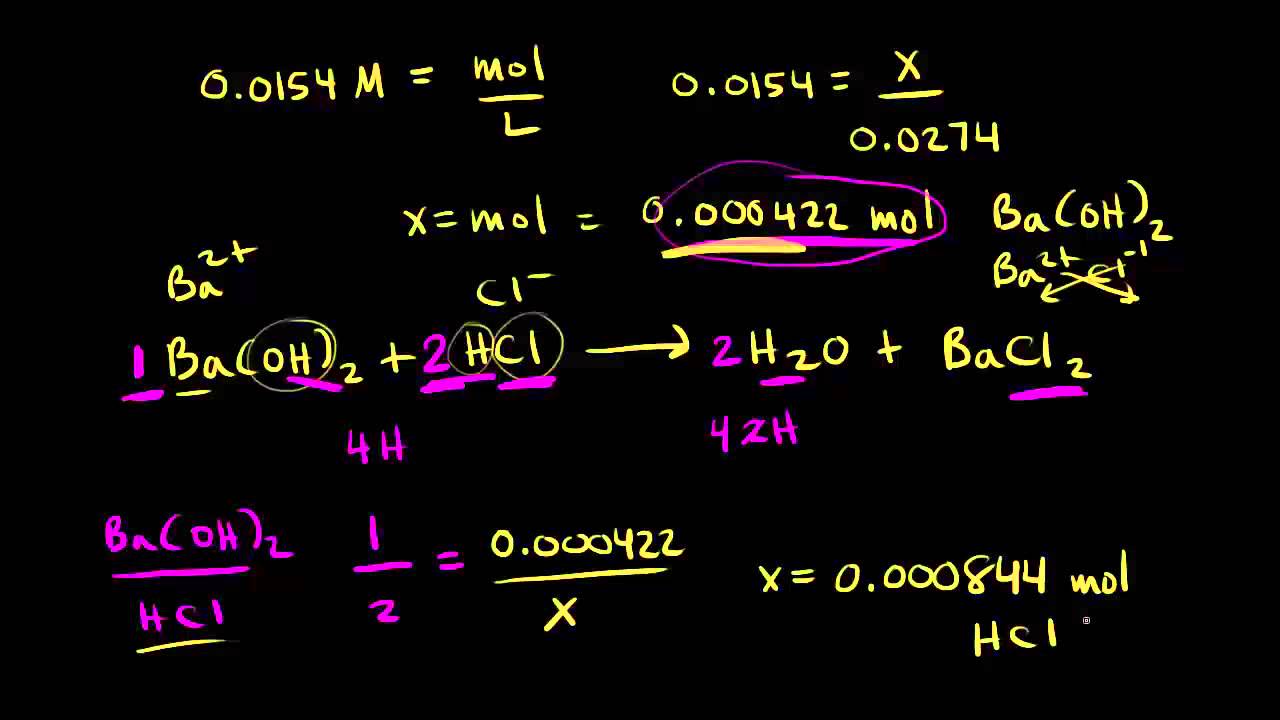PDF) CONDUCTOMETRIC TITRATION (DETERMINATION OF THE STRENGTH OF A SOLUTION OF HYDROCHLORIC ACID BY A STANDARD SOLUTION OF SODIUM-HYDROXIDE | Narasimha Chary - Academia.edu
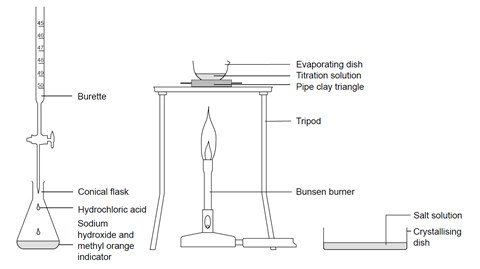
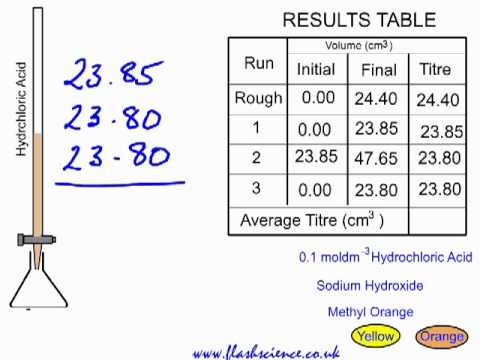
TITRATION Hydrochloric acid 0.1 mol/dm 3 Sodium hydroxide solution – concentration ? To determine the concentration of a solution of sodium hydroxide by. - ppt download

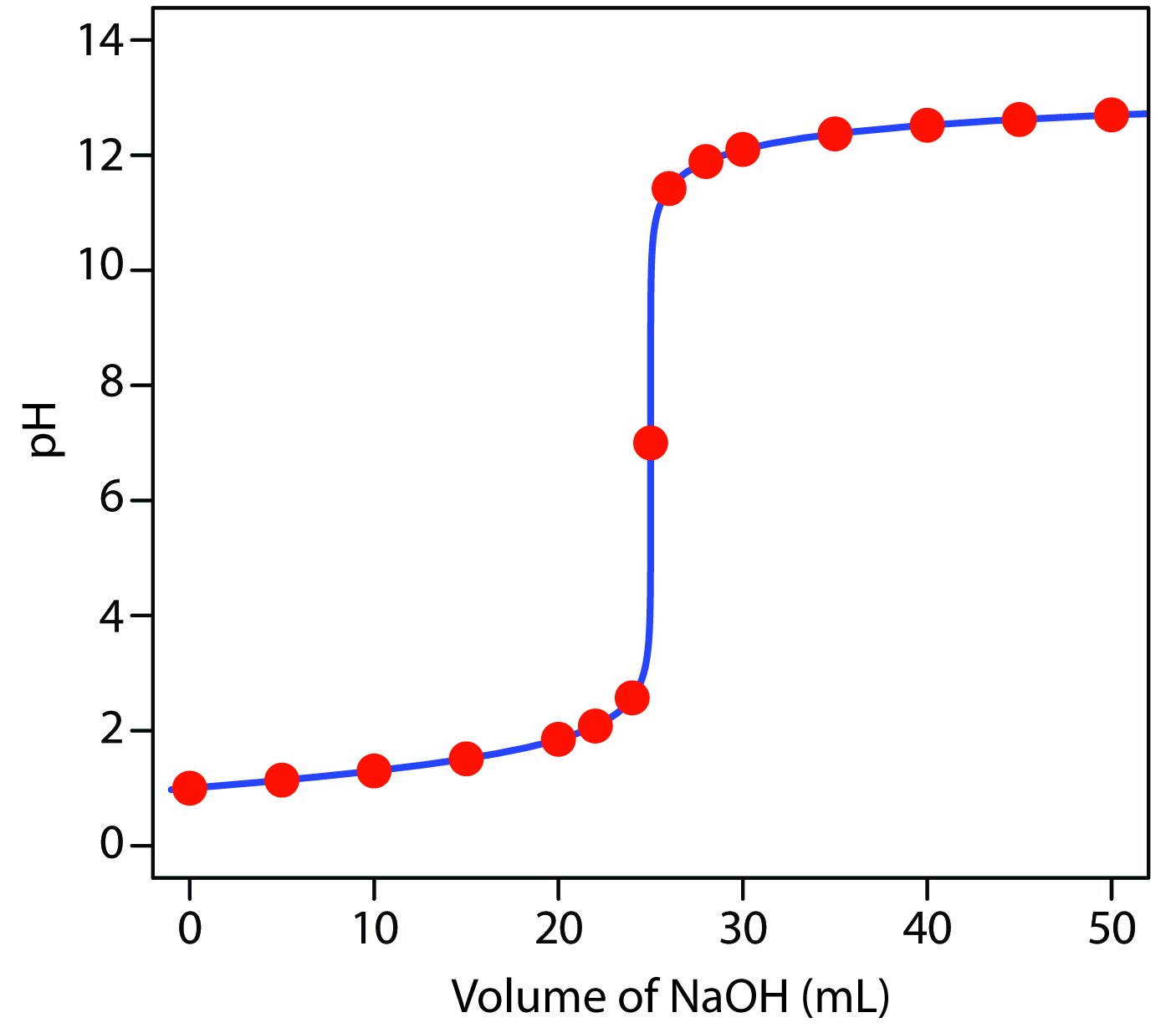
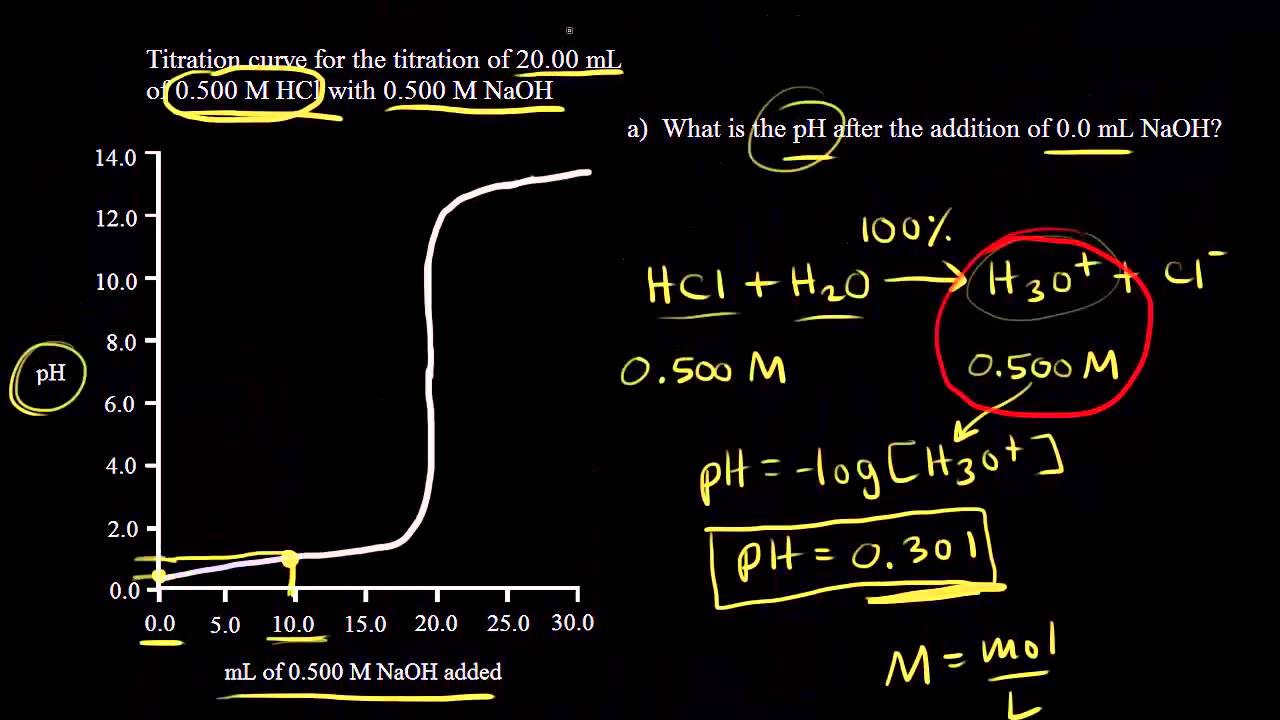
SOLVED: The following data is from conductometric titration of 10.00 mL aqueous solution containing mixture of acetic acid (CHCOOH) and hydrochloric acid (HCI) with 1.00 M solution of sodium hydroxide solution Volume